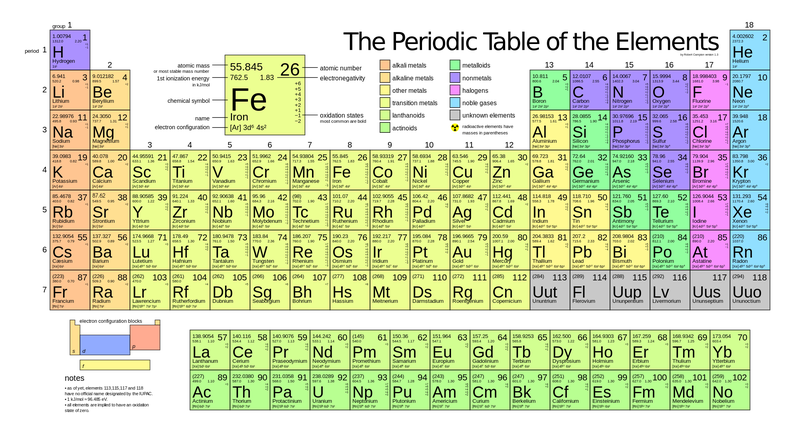
So the transition metals can also exhibit magnetic properties as a result of being able to have unpaired electrons, so if in the electron configuration you have paired electrons then we call that diamagnetic and if you actually have unpaired electrons, then we say that that element or compound exhibits paramagnetic properties. They usually have one oxidation state that they prefer, within the transition metals thought they can form numerous oxidation states for instance vanadium which is here atomic number 23, it can have an oxidation state of 2, 3, 4 and 5 so 2+, 3+, 4+ and 5+ and so all of those different oxidation states if you were to make a solution with vanadium you could know that the oxidation state was changing because the colors of the solution will change drastically. So transition metals can lose two s electrons to become 2+ions and they can also form multiple oxidation states so it's something different from the s-block elements and p-block elements. And so, when I say the electronic configurations I'm referring to the fact that if there are unpaired electrons, it causes the physical properties to be a little bit different within the family. However, there are differences in the physical properties and so those differences in physical properties are determined by the electronic configurations of the various elements that fall within these two groups. So across a period in the transition metals and the inner transition metals, remember periods are the rows, so going across the row there is little variation in the atomic size, in the electronegativity and in the ionization energy. So all these guys both the transition metals and the inner transition metals exhibit properties of the other metals that we see on the periodic table meaning they are good conductors of electricity, they have a nice luster quality to them and they're malleable which means they're soft and you kind of playable. Remember the d orbitals maximally hold 10 valence electrons and f orbitals hold a maximum of 14. Let's continue talking about the periodic table and more specifically let's talk about the properties of transition and inner transition metals, so we call that transition metals are groups 3 through 12 and they're right in the middle of the periodic table I'll point it out more materially and they have d orbitals and inner transition metals are located at the bottom of the periodic table and those guys have f orbitals.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
